|
For example, 6 12 C represents the carbon nucleus with six protons and six neutrons (or 12 nucleons). where X represents the chemical element, A is the mass number, and Z is the atomic number. A nucleus is represented symbolically by. The mass number is the sum of protons and neutrons. The total number of nucleons is the mass number, A. The atomic number of the atom is equal to the number of protons. The protium isotope is simply also known as hydrogen isotopes. So, the number of neutrons in protium is 0, deuterium is 1 and tritium is 2. The element uranium is made up of atoms with 92 protons and 92. $\Rightarrow Number\ of\ neutron = 3 - 1$ For example, hydrogen atoms, with their 1 proton and 1 electron, compose the element hydrogen. The number of neutrons in tritium is calculated as shown below. For example a hydrogen atom consists of one proton. $\Rightarrow Number\ of\ neutron = 2 - 1$ Because the number of protons in any atom is always equal to the number of electrons, atoms have no charge. The number of neutrons in deuterium is calculated as shown below. $\Rightarrow Number\ of\ neutron = 1 - 1$ The number of neutrons in Protium is calculated as shown below. $Number\ of\ neutron = mass\ number - atomic\ number$ The number of neutrons present in the nucleus is equal to the difference between the mass number of atom (M) and the atomic number (Z). In other words, the isotopes are explained as those species which have the same atomic number but differ in their mass number. The isotopes are defined as the species which contains the same number of protons but different numbers of neutrons. The atomic number of hydrogen is 1 and the standard atomic weight of the hydrogen is 1.008 which is obtained from the average mass of the three isotopes of the hydrogen. The nucleus of a deuterium atom, called a deuteron, contains one proton and one neutron, whereas the far more common protium has no neutrons in the nucleus. Hydrogen is the lightest element of all the chemical elements present in the periodic table. or D, also known as heavy hydrogen) is one of two stable isotopes of hydrogen (the other being protium, or hydrogen-1). Hydrogen is the chemical element present in the periodic table which is represented by the symbol H. All the three isotopes have similar atomic numbers but they differ from each other by the mass number. They are protium, deuterium, and tritium. iLooking at the picture, you can see there is only one electron in shell one.Hint: The atomic number of hydrogen is 1. That means there is only one electron in a normal hydrogen atom. In 1968, a Soviet K-129 mysteriously sank in the Pacific Ocean northwest of Hawaii, along with three nuclear missiles. for the element of HYDROGEN, you already know that the atomic number tells you the number of electrons. Once a shell is full, the next electron that is added must move to the next outer shell. It has only one electron and one proton because neutron cannot. (2-8-8) As you learn about elements with more than eighteen electrons you will find that shell three can hold more than eight. No, the hydrogen atom (Protium) does not contain neutron because of hydrogens small atomic size. Shell number one can only hold 2 electrons, shell two can hold 8, and for the first eighteen elements shell three can holds eight electrons. 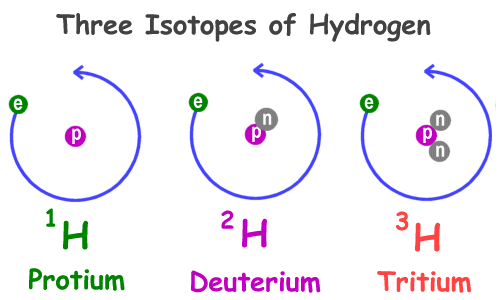
Electrons like to be in separate shells/orbitals. In an atom, the electrons spin around a central area called the nucleus. Each of those colored balls is an electron. Isotopes of atoms can have different numbers of neutrons. The relative atomic mass minus the atomic number tells you there are no neutrons in the nucleus of an average atom. Typical Unstable Isotopes Electrons and Electron Configuration The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. 
With the atomic number one, there is one proton in the nucleus and one electron orbiting the nucleus of a normal (electrically neutral) hydrogen atom. It tells you the mass of an average atom (relative atomic mass), how many pieces are inside, and where it should be placed on the periodic table. That box on the left has all of the information you need to know about one element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
